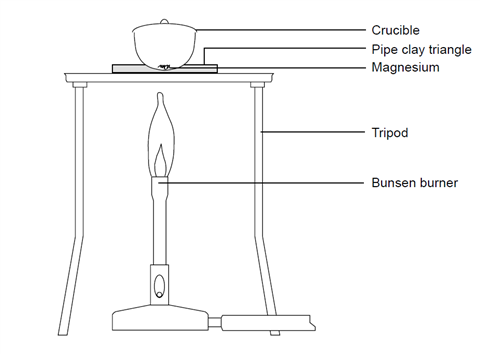
SOLVED: 2Mg(s) 02(g) 2MgO(s) molar massMg 24.30 g/mol molar masso2 31.98 g/mol molar massMgo 40.29 g/mol If 12.8 grams of oxygen completely react according to the provided equation and 31.2 grams of
UNCLASSIFIED AD NUMBER CLASSIFICATION CHANGES TO: FROM: LIMITATION CHANGES TO: FROM: AUTHORITY THIS PAGE IS UNCLASSIFIED
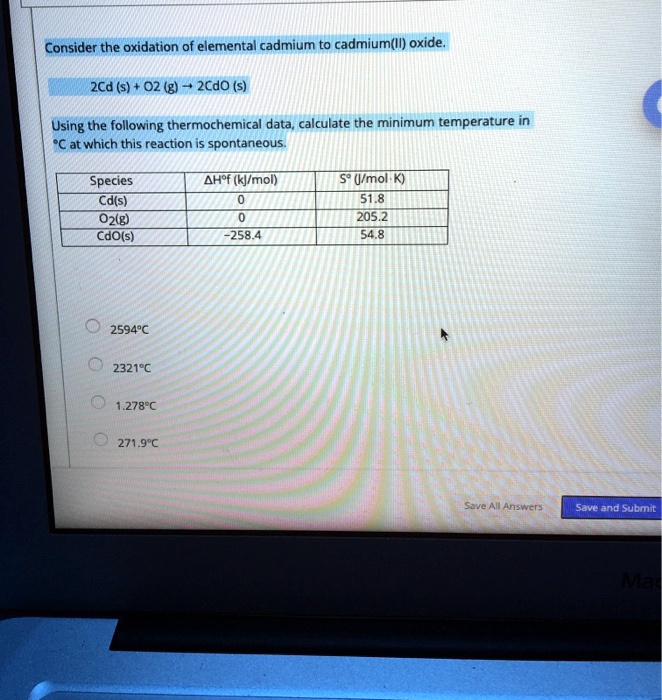
SOLVED: Consider the oxidation of elemental cadmium to cadmium(Il) oxide 2Cd (s) 02 (g) 2CdO (s) Using the following thermochemical data, calculate the minimum temperature in 'C at which this reaction spontaneous:

4pcs 2021 Year Carlomo 1/64 Alloy 9.5mm Wheels Super Smooth S Class 10.5mm Custom Tire 1:64 Model Car Tlv/ig/hw Modified Parts - Parts & Accs - AliExpress

SOLVED: Consider the following reaction at 298.0 K: Cu2s (s) + 02 (g) D 2 Cu (s) + SO2 (g) Kc = 0.0126 AHo = 30.87 kJ a) Calculate ASo in J/K

SOLVED: Write the complete, balanced chemical equation for the reaction between solid sodium and oxygen gas 2 Na (s) 02 (g) Naz0 (s) 02Na (s) 02 (g) 2 NaO (s) 2 Na (
UNCLASSIFIED AD NUMBER CLASSIFICATION CHANGES TO: FROM: LIMITATION CHANGES TO: FROM: AUTHORITY THIS PAGE IS UNCLASSIFIED

ACS Exam General Chemistry Equilibrium #4 - Consider this reaction 2C(s) + O2(g) -- 2CO(g) - YouTube

SOLVED: Given the thermochemical equation: Cu2O(s) Cu(s) 02 (g) AH = 448 kJImol Calculate the mass of copper generated when 1.47 x103 kJ is consumed in this reaction. Answer kg